MEDICAL DISCLAIMER & LEGAL NOTICE
| ⚠ READ BEFORE PROCEEDING |
This article is intended for educational and informational purposes only. It does not constitute medical advice, diagnosis, or treatment recommendations. GLP-1 medications are prescription drugs that require evaluation, prescription, and monitoring by a licensed healthcare professional. Individual results, side effects, and risks vary significantly. Do not self-medicate. If you experience a medical emergency, call 911 or your local emergency services immediately. |
The content presented in this article is based on peer-reviewed research, clinical trial data, and information from authoritative medical organizations including the U.S. Food and Drug Administration (FDA), the American Diabetes Association (ADA), and the Endocrine Society. All medical citations are noted with placeholders and should be verified against current literature.
Specific topics of caution include: (1) GLP-1 medications are not suitable for all individuals; (2) counterfeit medications sold online pose serious health risks; (3) stopping GLP-1 therapy abruptly can cause weight regain; (4) medications interact with other drugs — always disclose your full medication list to your prescriber.
| 💡 AFFILIATE DISCLOSURE |
This article may contain affiliate links to telehealth platforms, online pharmacies, and wellness programs. If you click a link and make a purchase or sign up, we may earn a commission at no additional cost to you. All recommendations are based on editorial merit, not compensation. Sponsored sections are clearly labelled. |
THE GLP-1 REVOLUTION
Beyond Weight Loss: What Ozempic, Mounjaro, Wegovy & Related Medications Can Really Do for Your Health
In 2026, a quiet revolution is reshaping modern medicine. The medications once celebrated as weight-loss 'miracle drugs' are revealing an entirely new identity, one that extends far beyond the bathroom scale. GLP-1 receptor agonists, the drug class that includes Ozempic (semaglutide), Wegovy (semaglutide), Mounjaro (tirzepatide), and Zepbound (tirzepatide), are emerging as some of the most powerful and versatile therapeutic tools physicians have ever encountered.
This is not hyperbole. Peer-reviewed clinical trials are demonstrating that these medications protect the heart from life-threatening cardiovascular events, shield the kidneys from diabetic damage, reduce systemic inflammation, show early promise in curbing addictive cravings, and may one day help slow cognitive decline. The story of GLP-1 drugs is no longer simply about losing weight. It is a story about rewriting how we understand, and treat metabolic disease at a cellular level.
Whether you are someone managing Type 2 diabetes, struggling with obesity, concerned about your heart health, or simply curious about the most talked-about medications of the decade, this comprehensive guide will give you the medically responsible, deeply researched, and humanely delivered information you deserve.
1. WHAT ARE GLP-1 DRUGS?
GLP-1 stands for Glucagon-Like Peptide-1, a naturally occurring hormone produced in the small intestine in response to food intake. Under normal physiological conditions, GLP-1 is released after you eat and performs several critical functions: it stimulates insulin secretion, suppresses glucagon (a hormone that raises blood sugar), slows gastric emptying, and signals the brain to register satiety, the feeling of fullness.
GLP-1 receptor agonists (GLP-1 RAs) are synthetic versions of this hormone, engineered to be far more potent and longer-lasting than the body's natural GLP-1, which degrades in minutes. By binding to GLP-1 receptors throughout the body, in the pancreas, brain, gut, heart, kidneys, and elsewhere; these medications produce a sustained cascade of metabolic benefits that researchers are only beginning to fully understand.
| 💡 DID YOU KNOW? |
The GLP-1 hormone was first isolated in 1983. It took nearly two decades of research before the first GLP-1 receptor agonist (exenatide / Byetta) was approved by the FDA in 2005 for diabetes. Today, newer agents like semaglutide and tirzepatide have transformed the field. |
The GLP-1 drug family now includes several agents approved for diabetes and/or obesity management: Ozempic (semaglutide, weekly injection), Wegovy (higher-dose semaglutide, weekly injection for obesity), Rybelsus (oral semaglutide, daily pill), Mounjaro (tirzepatide, weekly injection for diabetes), and Zepbound (tirzepatide, weekly injection for obesity).
2. HOW GLP-1 MEDICATIONS WORK IN THE HUMAN BODY

The mechanism of GLP-1 drugs is elegantly multi-systemic. Unlike earlier diabetes or weight-loss medications that targeted a single organ or pathway, GLP-1 receptor agonists act simultaneously across multiple organ systems.
The Pancreas: Blood Sugar Regulation
When GLP-1 binds to receptors on pancreatic beta cells, it triggers insulin secretion, but critically, only when blood glucose is elevated. This glucose-dependent mechanism greatly reduces the risk of hypoglycemia (dangerously low blood sugar) compared to older diabetes medications like sulfonylureas. Simultaneously, GLP-1 suppresses glucagon from alpha cells, preventing the liver from releasing stored glucose unnecessarily.
The Brain: Appetite and Reward
GLP-1 receptors are densely expressed in the hypothalamus and brainstem, regions that govern hunger, satiety, and reward. When activated, they reduce appetite, diminish food cravings, and promote a sustained feeling of fullness. Emerging research also suggests these brain pathways overlap with addiction circuits, which may explain why some patients report reduced cravings for alcohol, cigarettes, and compulsive eating behaviors.
The Gut: Slowed Digestion
GLP-1 slows gastric emptying, meaning food moves more slowly from the stomach into the small intestine. This blunts post-meal blood sugar spikes and contributes to prolonged satiety. While beneficial, this same mechanism can cause initial side effects of nausea and vomiting as the body adapts.
The Heart and Vessels: Cardiovascular Protection
GLP-1 receptors are present in cardiac muscle tissue and blood vessel walls. The activation of these receptors appears to reduce inflammation in arterial walls, improve endothelial function, lower blood pressure, and decrease harmful LDL cholesterol, all independent of weight loss alone.
The Kidneys: Renoprotection
GLP-1 receptors in the kidneys appear to reduce inflammatory signals and glomerular pressure, the microscopic filtration units of the kidney. In patients with diabetic kidney disease, this translates to measurably slower disease progression and reduced protein loss in urine (proteinuria).
3. THE SCIENCE BEHIND SEMAGLUTIDE AND TIRZEPATIDE
Semaglutide: The Molecule That Changed Medicine
Semaglutide (marketed as Ozempic, Wegovy, and Rybelsus) is a modified version of human GLP-1, engineered by Novo Nordisk with a fatty acid chain attached that binds to albumin in the bloodstream. This modification slows its degradation dramatically, extending its half-life to approximately 7 days, enabling once-weekly dosing. Semaglutide shares approximately 94% structural homology with native human GLP-1.
Clinical trial milestones for semaglutide include the SUSTAIN trials (cardiovascular outcomes in diabetes), the STEP trials (weight management), and the landmark SELECT trial (2023), which demonstrated a 20% reduction in major cardiovascular events among non-diabetic obese adults, a watershed moment that expanded semaglutide's clinical relevance far beyond diabetes.
Tirzepatide: The Dual-Agonist Breakthrough
Tirzepatide (Mounjaro for diabetes, Zepbound for obesity) represents the next generation of incretin-based therapy. Developed by Eli Lilly, it is a 'dual agonist'; activating both GLP-1 receptors AND GIP (Glucose-Dependent Insulinotropic Polypeptide) receptors simultaneously.
GIP is a sister incretin hormone that complements GLP-1 by further stimulating insulin release, promoting fat storage in healthy adipose tissue (reducing ectopic fat in the liver and muscles), and may enhance GLP-1 receptor sensitivity. The combination effect explains why tirzepatide achieves greater average weight loss (~20–22%) compared to semaglutide alone (~15%).
| 💡 CLINICAL MILESTONE |
The SURMOUNT-1 trial (2022) demonstrated that tirzepatide (15mg) achieved a mean body weight reduction of 22.5% at 72 weeks — the largest weight-loss effect ever observed in a randomized controlled trial of a pharmacological agent for obesity. [Citation: Jastreboff AM et al., NEJM, 2022] |
4. OZEMPIC vs MOUNJARO vs WEGOVY vs ZEPBOUND — FULL COMPARISON
With multiple GLP-1 medications now available, patients and prescribers often need to navigate important distinctions between them. Here is a comprehensive comparison of the most clinically relevant GLP-1 drugs available in 2026:
| Feature | Ozempic | Wegovy | Mounjaro | Zepbound | Rybelsus |
|---|---|---|---|---|---|
| Active Ingredient | Semaglutide | Semaglutide | Tirzepatide | Tirzepatide | Semaglutide (oral) |
| FDA Approval | Diabetes (2017) | Obesity (2021) | Diabetes (2022) | Obesity (2023) | Diabetes (2019) |
| Mechanism | GLP-1 RA | GLP-1 RA | GLP-1 + GIP | GLP-1 + GIP | GLP-1 RA |
| Avg. Weight Loss | ~10–15% | ~15–17% | ~20–22% | ~20–22% | ~5–10% |
| Route | Weekly injection | Weekly injection | Weekly injection | Weekly injection | Daily oral pill |
| Heart Benefit? | Yes (SUSTAIN-6) | Yes (SELECT trial) | Under study | Under study | Under study |
| Kidney Benefit? | Yes (FLOW trial) | Emerging data | Emerging | Emerging | Limited data |
| Monthly Cost (US) | ~$850–$950 | ~$1,200–$1,400 | ~$900–$1,050 | ~$500–$600* | ~$700–$850 |
*Zepbound pricing reflects available manufacturer savings cards as of 2026; actual cost varies. Prices listed reflect average US cash-pay rates before insurance or assistance programs.
| ⚠ IMPORTANT NOTE ON DRUG SELECTION |
The 'best' GLP-1 medication is highly individual and must be determined by a licensed physician. Factors including your specific health conditions, other medications, kidney and liver function, insurance coverage, and personal health goals all influence which agent is most appropriate for you. Never switch or start a GLP-1 drug without medical supervision. |
5. WHY GLP-1 DRUGS BECAME THE BIGGEST HEALTH TREND OF 2026

The meteoric rise of GLP-1 medications from niche diabetes treatments to global cultural phenomena is a story shaped by unprecedented clinical trial results, celebrity visibility, social media acceleration, and a world grappling with obesity as a chronic disease rather than a personal failing.
When the SELECT trial reported in 2023 that Wegovy reduced major cardiovascular events by 20% in non-diabetic obese patients, a population that has never had an effective pharmacological prevention tool, the medical world sat up and paid attention. When widespread anecdotal reports emerged of patients effortlessly losing 15–20% of their body weight, the cultural conversation exploded.
By 2025 and into 2026, GLP-1 prescriptions had grown by over 400% in three years. Manufacturers faced global shortages. Insurance battles raged. A black market of counterfeit medications emerged. Telehealth companies pivoted entirely to GLP-1 prescription services. The medications dominated medical conferences, financial headlines, and dinner table conversations alike.
But perhaps most significantly, the scientific community recognized that these drugs were not simply weight-loss tools; they appeared to be targeting the underlying biology of metabolic disease itself, opening therapeutic avenues in cardiology, nephrology, neurology, and addiction medicine that have energized an entire generation of researchers.
6. THE SHIFT BEYOND WEIGHT LOSS
The evolution of GLP-1 drugs from glycemic control agents to broad-spectrum metabolic medicines represents one of the most significant therapeutic pivots in modern pharmaceutical history. The shift occurred in identifiable stages:
Phase 1 (2005–2016):
GLP-1 agonists approved and used primarily for Type 2 diabetes management. Weight loss was noted as a welcome side effect.
Phase 2 (2017–2021):
Cardiovascular outcome trials revealed significant heart protection. FDA approvals for obesity (Wegovy) validated weight management as a primary therapeutic target.
Phase 3 (2022–2024):
Kidney protection data emerged. Early addiction research surfaced. Neurological and inflammatory applications entered serious clinical investigation.
Phase 4 (2025–Present):
Broad metabolic medicine paradigm. GLP-1 drugs are now being studied or used in NASH/NAFLD, sleep apnea, polycystic ovary syndrome (PCOS), addiction, Alzheimer's prevention, and inflammatory bowel disease.
This evolution reflects a fundamental truth about metabolic disease: it is not a collection of separate conditions but a deeply interconnected biological system. When you treat insulin resistance, you reduce cardiovascular risk. When you reduce systemic inflammation, you protect the kidneys and the brain. GLP-1 drugs, it turns out, are extraordinarily well-positioned to address the biological root of this system.
7. GLP-1 DRUGS AND TYPE 2 DIABETES
Type 2 diabetes remains the primary approved indication for most GLP-1 receptor agonists, and it is in this context that their benefits are most thoroughly documented. Approximately 422 million people globally live with diabetes (WHO, 2023), and managing glycemia while preventing complications, heart disease, kidney failure, blindness, neuropathy, remains the central clinical challenge.
Glycemic Control
GLP-1 receptor agonists reduce HbA1c (a 3-month blood sugar average) by an average of 1.0–2.0 percentage points, a clinically meaningful reduction comparable to or exceeding most other glucose-lowering medications. Crucially, the glucose-dependent mechanism of insulin stimulation means the risk of hypoglycemia is substantially lower than with insulin or sulfonylureas.
Beta Cell Preservation
Emerging evidence from preclinical and some clinical studies suggests that GLP-1 drugs may actually preserve beta cell function and mass over time, slowing the progression of diabetes rather than simply managing its symptoms. This finding, if confirmed in long-term human trials, would be paradigm-shifting. [Citation: Drucker DJ, Cell Metabolism, 2022 — Placeholder]
Weight Loss Enhances Diabetes Outcomes
In patients with Type 2 diabetes, the weight loss achieved with GLP-1 drugs produces compounding benefits: improved insulin sensitivity, reduced fatty liver infiltration, lower cardiovascular risk, and in some patients, partial or full remission of diabetes, particularly when treated early in the disease course.
8. CARDIOVASCULAR BENEFITS AND HEART DISEASE PREVENTION
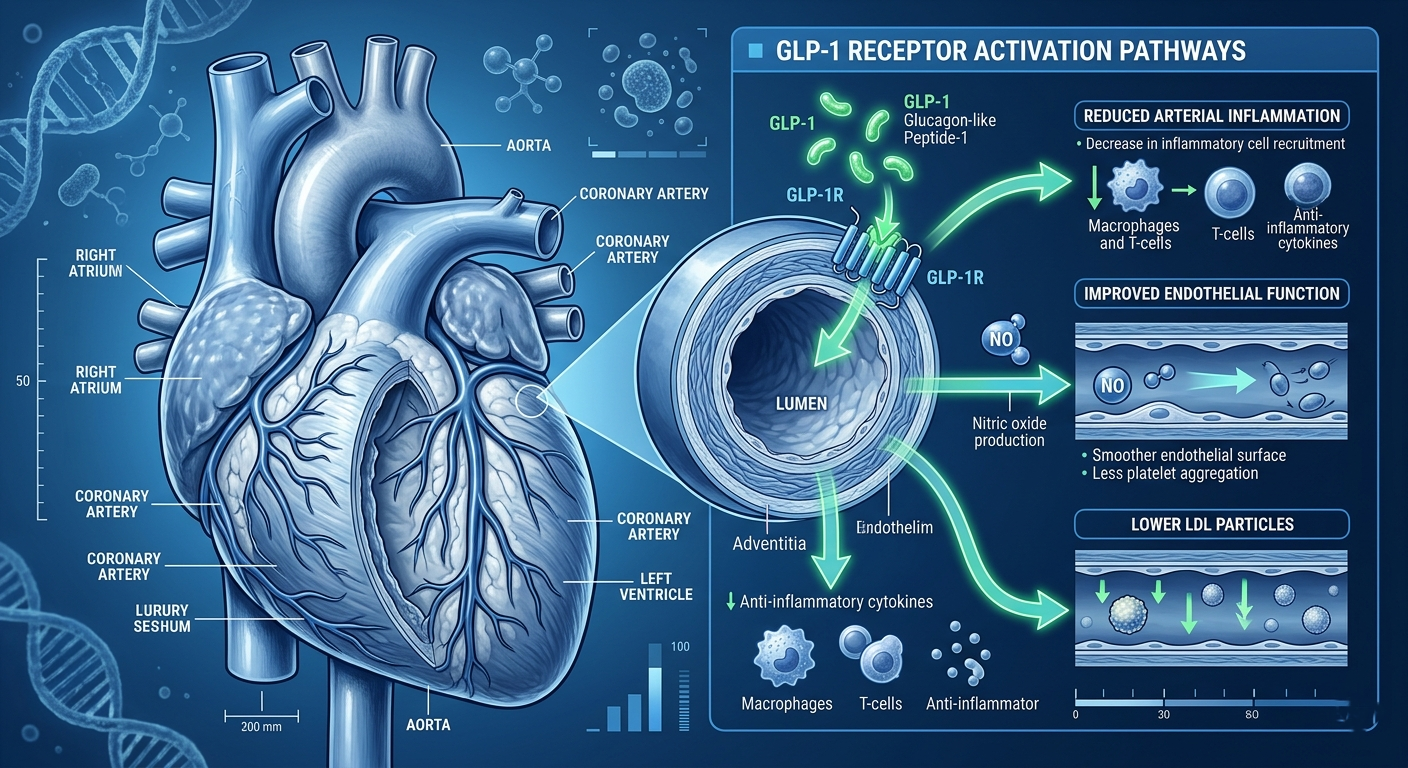
Perhaps the most clinically transformative finding in the GLP-1 story has been the discovery of robust cardiovascular protection. Heart disease remains the leading cause of death globally, and obesity and Type 2 diabetes are among its most powerful risk factors.
The SELECT Trial: A Historic Breakthrough
Published in the New England Journal of Medicine in 2023, the SELECT trial enrolled over 17,600 obese or overweight adults with established cardiovascular disease but without diabetes. Participants treated with semaglutide 2.4mg weekly (Wegovy) experienced a 20% reduction in the composite risk of cardiovascular death, non-fatal heart attack, and non-fatal stroke compared to placebo, over an average follow-up of 39 months. [Citation: Lincoff AM et al., NEJM, 2023]
This finding was significant for multiple reasons: it confirmed cardiovascular benefit in non-diabetic patients, it suggested the benefit extended beyond weight loss alone (anti-inflammatory and direct vascular effects were implicated), and it positioned GLP-1 drugs as potential first-line cardiovascular prevention tools.
Earlier Cardiovascular Trials
The SUSTAIN-6 trial (2016) and LEADER trial (liraglutide, 2016) were among the first cardiovascular outcome trials to demonstrate that GLP-1 drugs reduced major adverse cardiovascular events (MACE) in high-risk diabetes patients — reshaping prescribing guidelines globally.
Proposed Cardiovascular Mechanisms
Direct anti-inflammatory effects in coronary arterial walls
Improved endothelial function (the lining of blood vessels)
Reduction in blood pressure (systolic BP decreases of 2–5 mmHg)
Modest reduction in LDL cholesterol and triglycerides
Reduced progression of atherosclerotic plaque
Lowered levels of high-sensitivity C-reactive protein (hs-CRP), a key inflammation marker
9. KIDNEY PROTECTION AND METABOLIC HEALTH
Diabetic kidney disease (diabetic nephropathy) affects approximately 40% of people with Type 2 diabetes and is the leading cause of chronic kidney disease and kidney failure worldwide. The kidneys are exquisitely sensitive to the metabolic consequences of sustained hyperglycemia, hypertension, and inflammation.
The FLOW Trial: Landmark Kidney Data
The FLOW trial, published in 2024, evaluated semaglutide 1.0mg in patients with Type 2 diabetes and chronic kidney disease. Results showed a 24% reduction in the composite kidney endpoint (worsening kidney function, kidney failure, or kidney-related death) and a 29% reduction in cardiovascular death, establishing semaglutide as the first GLP-1 drug with evidence-based kidney protection in this population. [Citation: Perkovic V et al., NEJM, 2024 — Placeholder]
Mechanisms of Kidney Protection
Reduction of intraglomerular pressure — the blood pressure within kidney filtration units
Anti-inflammatory effects within kidney tubules
Reduced proteinuria (protein in urine — a marker of kidney damage)
Blood pressure lowering (both systemic and renal vascular)
Metabolic improvements that reduce oxidative stress in kidney tissue
| 💡 METABOLIC LIVER DISEASE (NAFLD/NASH) |
Non-alcoholic fatty liver disease (NAFLD) and its inflammatory progression, NASH, affect an estimated 25% of the global adult population and represent a growing liver disease epidemic. Early clinical data on semaglutide in NASH patients has been promising, with a Phase 3 trial (ESSENCE) showing significant liver fat reduction and histological improvement. GLP-1 drugs may become a cornerstone of NAFLD/NASH therapy. |
10. EMERGING RESEARCH ON ADDICTION AND CRAVING REDUCTION
One of the most surprising, and scientifically fascinating, emerging applications of GLP-1 drugs is their apparent ability to reduce addictive cravings and compulsive behaviors. This finding, which emerged largely from patient self-reports before being taken up by researchers, is now the subject of serious clinical investigation.
The 'Quiet Food Noise' Phenomenon
Many patients on GLP-1 medications report that the constant mental preoccupation with food, the intrusive thoughts about eating, the craving loops, the compulsive snacking, simply becomes quieter. This effect, described colloquially as 'food noise going silent,' correlates with GLP-1 receptor activation in the mesolimbic dopaminergic system: the brain's reward and motivation circuitry.
Alcohol and Substance Use
Multiple observational studies and preclinical animal data suggest that GLP-1 receptor activation reduces alcohol consumption, cocaine self-administration, and nicotine dependence behaviors. A 2024 retrospective study of patients on semaglutide showed a significant reduction in alcohol use disorder diagnoses compared to matched controls not on GLP-1 drugs. Randomized controlled trials are now underway. [Citation: Klausen MK et al., JCI Insight, 2022 — Placeholder]
Gambling and Compulsive Behaviors
Early case series and patient reports suggest possible reductions in compulsive gambling, compulsive shopping, and binge eating disorder behaviors. These signals are preliminary but mechanistically plausible given the GLP-1 system's role in reward valuation and impulsivity modulation.
| ⚠ IMPORTANT CAUTION ON ADDICTION USE |
GLP-1 drugs are NOT currently FDA-approved for treating addiction, alcohol use disorder, or any substance use disorder. Prescribing for these purposes is off-label. The research is promising but preliminary. Anyone struggling with addiction should consult an addiction medicine specialist for evidence-based treatment options. |
11. POSSIBLE LINKS TO BRAIN HEALTH AND ALZHEIMER'S PREVENTION

Among the most exciting, and most speculative, frontiers for GLP-1 research is the potential neuroprotective role of these medications in cognitive health and Alzheimer's disease prevention.
GLP-1 receptors are expressed throughout the central nervous system, including in areas critical to memory, executive function, and neuronal survival.
The Neurological Rationale
Alzheimer's disease and Type 2 diabetes share striking biological features: insulin resistance, neuroinflammation, impaired glucose metabolism in the brain, amyloid-beta accumulation, and tau protein dysregulation. Some researchers have proposed that Alzheimer's may represent a form of 'brain insulin resistance'; a provocative hypothesis that has been termed 'Type 3 diabetes.' If GLP-1 drugs can reverse systemic insulin resistance, the question naturally follows: can they do the same in the brain?
Early Clinical Evidence
Liraglutide (Victoza) was evaluated in the ELAD trial for mild Alzheimer's disease. Results published in 2024 showed a significant slowing of brain volume loss in treated patients and hints of cognitive stabilization, though the trial did not meet its primary endpoint. [Citation: ELAD Trial, Lancet, 2024 — Placeholder]
Larger trials evaluating semaglutide in Alzheimer's prevention (EVOKE trials, ALZHEIMER'S ASSOCIATION sponsored studies) are now underway, with results expected in 2026–2028.
Other Neurological Applications Under Study
Parkinson's disease: GLP-1 may protect dopaminergic neurons
Traumatic brain injury recovery
Depression and anxiety (overlapping neuroinflammatory mechanisms)
Multiple sclerosis (anti-neuroinflammatory properties)
12. GLP-1 DRUGS AND INFLAMMATION REDUCTION
Chronic low-grade inflammation, sometimes called 'metaflammation' or metabolic inflammation, is a central driver of virtually every major chronic disease: Type 2 diabetes, cardiovascular disease, cancer, Alzheimer's, autoimmune conditions, and aging itself. GLP-1 drugs appear to exert meaningful anti-inflammatory effects through multiple pathways.
Anti-Inflammatory Mechanisms
Suppression of NF-κB signaling — a master regulator of inflammatory gene expression
Reduction of pro-inflammatory cytokines (TNF-alpha, IL-6, IL-1beta)
Lowering of C-reactive protein (CRP) — a widely used inflammation biomarker
Reduction of macrophage infiltration into adipose tissue (fat tissue inflammation)
Mitigation of endoplasmic reticulum stress in metabolic tissues
Separately from metabolic effects, these anti-inflammatory properties may contribute independently to the cardiovascular, renal, hepatic, and neurological benefits observed in clinical trials, making anti-inflammation a mechanistic thread that runs through the entire GLP-1 story.
13. BENEFITS FOR OBESITY AND INSULIN RESISTANCE

Obesity is now recognized by every major medical organization as a chronic, complex, relapsing, and neurobiological disease, not a character flaw or lifestyle failure. The mechanisms of obesity involve genetic predisposition, epigenetic programming, environmental factors, gut microbiome composition, hormonal dysregulation, and brain reward circuitry, all of which GLP-1 drugs address to varying degrees.
How GLP-1 Drugs Produce Weight Loss
Reduced caloric intake through appetite suppression (primary mechanism)
Slowed gastric emptying prolonging fullness after eating
Reduced hedonic eating — eating for pleasure or reward rather than hunger
Possible increase in resting energy expenditure (metabolic rate) — under investigation
Preservation of lean muscle mass compared to caloric restriction alone (though muscle loss is a concern with rapid loss)
Insulin Resistance Reversal
Excess adiposity, particularly visceral (abdominal) fat, drives insulin resistance by releasing inflammatory signals and fatty acids that impair insulin signaling in muscle, liver, and fat tissue. As GLP-1 drugs reduce body fat mass, insulin sensitivity improves substantially. In many patients, this improvement is measurable within weeks, independent of the degree of weight lost.
14. COMMON SIDE EFFECTS AND SAFETY CONCERNS
GLP-1 medications are generally well-tolerated, but like all pharmacological agents, they carry a profile of adverse effects that patients and prescribers must understand. Most side effects are gastrointestinal and occur predominantly during the dose-escalation phase as the body adjusts to the medication.
| Common (>10%) | Less Common (1–10%) | Rare but Serious (<1%) |
|---|---|---|
| Nausea | Diarrhea | Pancreatitis |
| Vomiting | Constipation | Gallbladder disease |
| Decreased appetite | Bloating / gas | Thyroid C-cell tumors (rodent data) |
| Fatigue (initial) | Heartburn / reflux | Kidney injury (dehydration) |
| Injection site redness | Hair thinning (rapid loss) | Severe allergic reaction |
| Mild headache | Low blood sugar (w/ insulin) | Suicidal ideation (rare, under review) |
The majority of gastrointestinal side effects are transient, peaking in the first 4–8 weeks and diminishing significantly thereafter. Starting at low doses and escalating gradually (as prescribed) substantially reduces the severity of initial side effects.
Managing Side Effects Effectively
Eat smaller, more frequent meals during the adjustment period
Avoid high-fat, spicy, or heavily processed foods that exacerbate nausea
Stay well-hydrated — dehydration can worsen nausea and increase kidney risk
Take injections at a consistent time each week
Do not skip doses to avoid side effects without consulting your prescriber
Report persistent vomiting, severe abdominal pain, or unusual symptoms to your doctor immediately
15. LONG-TERM RISKS AND UNKNOWNS
Because GLP-1 receptor agonists represent a relatively young drug class — with the first approvals in 2005 — comprehensive long-term safety data spanning decades remains limited. This is not a reason to avoid these medications when indicated, but it is a reason for ongoing medical supervision and transparency with patients about what is and is not yet known.
Thyroid Concerns
Rodent studies showed dose-dependent increases in thyroid C-cell tumors with GLP-1 agonists. While this finding has NOT been replicated in human studies or epidemiological data to date, GLP-1 drugs carry an FDA black-box warning for patients with personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia type 2 (MEN 2). Long-term thyroid cancer surveillance studies are ongoing.
Pancreatitis Risk
Some earlier pharmacovigilance data suggested a possible association between GLP-1 drugs and pancreatitis. However, multiple large cardiovascular outcome trials and meta-analyses have not confirmed a statistically significant increased risk. Nonetheless, patients with a history of pancreatitis should use these medications with caution and under close supervision.
Muscle Loss (Sarcopenia)
Rapid weight loss of any cause can produce loss of lean muscle mass alongside fat. This is a legitimate concern, particularly in older adults, as muscle loss increases fall risk and metabolic fragility. Adequate dietary protein and resistance exercise are critical protective strategies (see Section 23).
Mental Health Signals
In 2023, the FDA and European Medicines Agency (EMA) initiated a review of reports of suicidal ideation and self-harm in patients using GLP-1 drugs. After review of available data, regulatory agencies concluded that the evidence did not support a causal link at that time, though monitoring continues. Patients with a history of depression or suicidal ideation should be monitored closely.
Rebound Weight Regain
Multiple studies have demonstrated that when GLP-1 therapy is discontinued, appetite and body weight tend to return toward baseline within 6–12 months. This reflects the biological reality of obesity as a chronic disease requiring ongoing management — not a failure of the medication or the patient.
16. MENTAL HEALTH AND EMOTIONAL EFFECTS
The psychological dimensions of using GLP-1 medications deserve compassionate, nuanced attention. Weight and eating are deeply entangled with identity, emotion, trauma, and social experience. The profound appetite suppression and rapid body changes associated with GLP-1 drugs can evoke complex emotional responses.
Positive Psychological Effects Reported
Reduced anxiety around food and eating situations
Improved body image and self-confidence as weight changes
Greater energy levels and physical mobility enhancing mood
Reduced depression scores observed in several clinical studies
Enhanced sense of agency and control over health
Emotional Challenges to Anticipate
Grief or identity adjustment as relationship with food changes
Difficulty eating socially or enjoying traditional celebratory foods
Anxiety about stopping medication and weight regain
Pressure from social networks to justify or explain medication use
Possible exposure to fatphobic or stigmatizing messaging (from both 'pro' and 'anti' medication perspectives)
| 💡 A NOTE ON EMOTIONAL SUPPORT |
| If you are using or considering GLP-1 therapy, working with a therapist or behavioral health specialist — ideally one with experience in weight and body image concerns — can significantly improve your long-term outcomes and overall wellbeing. Medication works best as part of a holistic care approach. |
17. WHO SHOULD AND SHOULD NOT USE GLP-1 MEDICATIONS
GLP-1 medications are not appropriate for everyone. The following eligibility checklist is a general guide — your individual suitability must be determined by a licensed healthcare provider based on your complete medical history.
| ✅ May Be Eligible | ❌ Use With Caution or Avoid |
|---|---|
| BMI ≥ 30 (or ≥ 27 with weight-related complication) | History of medullary thyroid carcinoma (MTC) |
| Type 2 diabetes not controlled by lifestyle alone | Multiple Endocrine Neoplasia type 2 (MEN 2) |
| Established cardiovascular disease (heart attack, stroke history) | Personal or family history of pancreatitis |
| Chronic kidney disease with albuminuria | Severe kidney or liver impairment |
| Non-alcoholic fatty liver disease (NAFLD/NASH) | Pregnancy or breastfeeding |
| Metabolic syndrome with two or more markers | Children under 12 (most formulations) |
18. THE DANGERS OF SELF-MEDICATION WITHOUT MEDICAL SUPERVISION
| ⚠ DO NOT SELF-MEDICATE WITH GLP-1 DRUGS |
| GLP-1 medications are prescription-only substances requiring medical evaluation before use. Self-administering these drugs without a prescription, purchasing them from unverified online sources, or using doses not prescribed to you can result in serious harm, including severe dehydration, pancreatitis, hypoglycemia (in combination with other medications), dangerous cardiovascular events, and exposure to counterfeit or contaminated products. |
The explosion in GLP-1 demand has unfortunately generated a parallel market of dangerous self-medication practices. Social media forums share injection protocols and compounded formulations without medical oversight. Patients with contraindications unknowingly expose themselves to serious risks. Rapid dose escalation outside of supervised titration schedules has led to emergency hospitalizations.
Medical supervision throughout GLP-1 therapy is not bureaucratic red tape — it is clinically essential. A prescribing clinician will: evaluate your complete health history and current medications; select the appropriate GLP-1 agent and starting dose; guide dose escalation on a medically sound schedule; monitor for adverse effects including pancreatitis, kidney function, and thyroid parameters; provide dietary and lifestyle guidance to maximize results; and manage any complications that arise.
19. FAKE OZEMPIC AND ONLINE DRUG SCAMS
| ⚠ COUNTERFEIT GLP-1 DRUGS — A GLOBAL HEALTH EMERGENCY |
| The World Health Organization (WHO) issued a global alert in 2024 regarding counterfeit semaglutide products circulating in multiple countries, including the United States, United Kingdom, and across Europe. These fake products have been found to contain incorrect doses, impurities, non-sterile compounds, insulin (which can cause life-threatening hypoglycemia), and in some cases, entirely different substances. Hospitalisations and deaths have been reported in connection with counterfeit GLP-1 products. |
How to Protect Yourself
Only obtain GLP-1 medications through a licensed pharmacy with a valid prescription
Use manufacturer-verified pharmacies or telehealth platforms with licensed dispensing partners
Be extremely wary of online sellers offering Ozempic, Wegovy, or Mounjaro at dramatically reduced prices
Verify the physical appearance of your medication against manufacturer documentation
Report suspicious products to the FDA MedWatch program (www.fda.gov/medwatch)
If purchasing compounded semaglutide, ensure it comes from an FDA-registered 503B outsourcing facility
| 💡 COMPOUNDED SEMAGLUTIDE — WHAT YOU NEED TO KNOW |
| During the period of FDA-declared shortage, compounding pharmacies legally produced semaglutide formulations. As of 2025, the FDA declared the shortage resolved, creating legal pressure to discontinue most compounded semaglutide. However, the regulatory landscape is evolving. Always consult your prescriber and ensure any compounded formulation comes from a licensed, regulated facility. |
20. COST, INSURANCE, AND ACCESSIBILITY CHALLENGES
One of the most significant barriers to GLP-1 therapy is cost. These medications rank among the most expensive in the world, and insurance coverage remains inconsistent and often insufficient.
Average Monthly Costs (US, 2026)
Ozempic (semaglutide 0.5–1.0mg): ~$850–$950/month without insurance
Wegovy (semaglutide 2.4mg): ~$1,200–$1,400/month without insurance
Mounjaro (tirzepatide): ~$900–$1,050/month without insurance
Zepbound (tirzepatide, savings card): ~$499–$600/month without insurance
Rybelsus (oral semaglutide): ~$700–$850/month without insurance
Insurance Coverage Challenges
Medicare Part D historically excluded coverage for weight-loss medications, though the Treat and Reduce Obesity Act (TROA), reintroduced in Congress, continues to be debated. Many commercial insurance plans cover GLP-1 drugs for diabetes but not obesity, even when the same medication (different dose) is approved for both. The disparity reflects continued stigmatization of obesity as a medical condition.
Cost-Reduction Strategies
Novo Nordisk NovoCare and Eli Lilly LillyDirect manufacturer savings programs
Patient assistance programs (PAPs) for qualifying low-income patients
GoodRx, NeedyMeds, and other prescription discount platforms
Telehealth platforms that negotiate compounded formulations (with caveats — see Section 19)
Advocating with your employer's HR for insurance benefit inclusion
21. LIFESTYLE CHANGES THAT IMPROVE GLP-1 RESULTS
GLP-1 medications are powerful, but they are not magic. Clinical trials consistently show that patients who combine GLP-1 therapy with intentional lifestyle changes achieve superior outcomes in weight management, metabolic health, and long-term sustainability.
LIFESTYLE OPTIMIZATION CHECKLIST |
|
22. DIET AND NUTRITION WHILE USING GLP-1 DRUGS

Because GLP-1 medications dramatically reduce appetite, patients often consume significantly fewer calories — sometimes too few. Without intentional dietary planning, this can result in nutritional deficiencies, loss of lean muscle mass, fatigue, and long-term metabolic complications.
Nutritional Priorities on GLP-1 Therapy
Protein First: Every meal should prioritize lean protein (chicken, fish, eggs, tofu, Greek yogurt, legumes). Protein supports muscle preservation, satiety, and metabolic rate. Target: 25–40g protein per meal.
Micronutrient Density: With reduced caloric intake, every calorie counts nutritionally. Prioritize vegetables, whole grains, nuts, seeds, and legumes over empty-calorie foods.
Avoid Trigger Foods: Foods high in fat and sugar can worsen GLP-1 side effects (nausea, reflux). Greasy, fried, or heavily processed foods are particularly problematic during the adjustment phase.
Small, Frequent Meals: Three moderate meals with small protein-rich snacks often work better than two large meals when appetite is suppressed.
Hydration: Reduced food intake can reduce fluid consumption. Conscious hydration is essential, particularly during nausea episodes.
Supplementation: Many clinicians recommend a multivitamin plus vitamin D, calcium, magnesium, and omega-3 supplementation during GLP-1 therapy — discuss with your prescriber.
| 💡 FOODS TO MINIMIZE ON GLP-1 THERAPY |
| Alcohol, high-fat fried foods, carbonated beverages (worsen bloating and nausea), very high-fiber foods (during early adjustment), and ultra-processed snacks. These foods either worsen side effects, undermine metabolic progress, or displace essential nutrients. |
23. EXERCISE AND MUSCLE PRESERVATION STRATEGIES
Muscle mass is metabolically precious. It drives resting energy expenditure, supports physical function, protects bone density, improves insulin sensitivity, and dramatically affects long-term weight maintenance. Rapid weight loss — which GLP-1 drugs can produce — carries a risk of muscle loss alongside fat loss. Exercise is the most powerful countermeasure available.
Resistance Training: The Non-Negotiable
Resistance (strength) training — using weights, resistance bands, or bodyweight exercises — is the single most effective strategy for preserving and building muscle during GLP-1-induced weight loss. Aim for: 2–3 sessions per week, targeting all major muscle groups; progressive overload (gradually increasing resistance or repetitions over time); compound movements (squats, deadlifts, rows, presses) that recruit multiple muscle groups simultaneously; and rest and recovery between sessions.
Aerobic Exercise for Cardiovascular and Metabolic Health
Aerobic exercise — walking, cycling, swimming, jogging — enhances the cardiovascular and metabolic benefits of GLP-1 drugs. A minimum of 150 minutes of moderate-intensity aerobic activity per week is recommended. Patients new to exercise should start slowly and progress gradually with guidance.
Protein Intake + Exercise: A Synergistic Strategy
The combination of adequate protein intake (1.2–1.6g per kg body weight per day) and regular resistance exercise is consistently associated with the best muscle preservation outcomes during weight-loss therapy. Neither strategy alone is as effective as the combination.
24. TELEHEALTH AND ONLINE PRESCRIPTION PLATFORMS

The rise of GLP-1 drugs has coincided with — and accelerated — a revolution in telehealth, making access to metabolic medicine consultations, prescriptions, and monitoring more accessible than ever before. However, the telehealth landscape for GLP-1 drugs is highly variable in quality and regulatory compliance.
Leading Telehealth Platforms for GLP-1 Access (2026)
[AFFILIATE DISCLOSURE: The following platforms are listed for informational purposes. This article may receive affiliate compensation for referrals. All platforms have been editorially evaluated.]
| Platform | Services | GLP-1 Access | Starting Cost |
|---|---|---|---|
| Hims & Hers | Men's & Women's health | Compounded semaglutide | ~$199/mo |
| Ro Body | Weight management program | Ozempic / Wegovy prescription | ~$99/mo + Rx |
| Found | Metabolic health coaching | Full GLP-1 program | ~$99/mo + Rx |
| Sequence | Obesity medicine specialists | Insurance navigation + GLP-1 | ~$99/mo |
| Noom Med | Behavior + medication | Integrated weight-loss plan | ~$149/mo |
| Calibrate | Metabolic reset program | 1-year metabolic program + Rx | ~$149/mo |
What to Look for in a Telehealth GLP-1 Provider
Board-certified physicians or nurse practitioners with obesity or endocrinology expertise
Comprehensive medical intake (not just a brief questionnaire)
Ongoing monitoring and follow-up appointments
Transparent prescribing practices (original branded medications preferred over unverified compounds)
Licensed pharmacy partners and prescription verification
Clear escalation protocols for side effects and emergencies
No 'subscription trap' cancellation policies — you should be able to pause or stop easily
25. FREQUENTLY ASKED QUESTIONS (FAQ)
The following FAQ section is formatted for featured-snippet optimization and provides medically responsible answers to the most commonly searched GLP-1 drug questions.
| Q: Is Ozempic the same as Wegovy? |
A: Both contain semaglutide, but they differ in approved doses and indications. Ozempic (up to 2.0mg) is approved for Type 2 diabetes. Wegovy (2.4mg) is approved for chronic weight management. The higher Wegovy dose produces greater average weight loss. They are manufactured differently and are not interchangeable without physician guidance. |
| Q: How long does it take to see results on a GLP-1 drug? |
A: Most patients notice appetite reduction within the first 1–2 weeks. Meaningful weight loss is typically apparent by weeks 4–8. Maximum weight loss is usually achieved at 52–72 weeks of consistent therapy. Glycemic improvements in diabetic patients can appear within days of initiating therapy. |
| Q: Can I stop taking GLP-1 drugs if I reach my goal weight? |
A: This is an individual decision to make with your prescriber. Research shows that appetite and weight tend to return toward baseline within 6–12 months of stopping therapy, reflecting the chronic disease nature of obesity. Some patients successfully maintain with lower-dose maintenance therapy; others work with their physician to taper off with a robust lifestyle maintenance plan in place. |
| Q: Are GLP-1 drugs safe for people over 65? |
A: GLP-1 drugs can be used in older adults with appropriate monitoring. Key considerations include: muscle loss risk (resistance exercise is especially important), kidney function monitoring, dehydration risk (nausea and vomiting can impair hydration), and potential interactions with other medications common in older adults. Prescribers typically start with lower doses and escalate more slowly. |
| Q: Will insurance cover my GLP-1 medication? |
A: Coverage varies dramatically by insurer, employer, and indication. GLP-1 drugs prescribed for Type 2 diabetes are more commonly covered than those prescribed for obesity alone. Contact your insurer directly, ask your prescriber's office for prior authorization support, and investigate manufacturer patient assistance programs (NovoCare, LillyDirect) if coverage is denied. |
| Q: What is the difference between semaglutide and tirzepatide? |
A: Semaglutide (Ozempic, Wegovy) is a pure GLP-1 receptor agonist. Tirzepatide (Mounjaro, Zepbound) is a dual GLP-1/GIP receptor agonist, which produces greater average weight loss (~20–22% vs ~15%) due to its additional GIP mechanism. However, individual responses vary; some patients achieve excellent results with semaglutide. The right choice depends on your medical profile and prescriber judgment. |
| Q: Can GLP-1 drugs cure diabetes? |
A: GLP-1 drugs are not a cure for Type 2 diabetes, but they can produce partial or full remission in some patients — particularly those in the earlier stages of disease with significant residual beta cell function. Weight loss achieved with GLP-1 therapy meaningfully reduces insulin requirements and can normalize blood glucose. However, discontinuing the medication often leads to some return of hyperglycemia. |
| Q: Are there non-injectable GLP-1 options? |
A: Yes. Rybelsus (oral semaglutide) is an FDA-approved daily oral pill for Type 2 diabetes. It produces less weight loss than injectable semaglutide but is an option for patients who strongly prefer an oral route. An oral version of tirzepatide (orforglipron) is in late-stage clinical trials and may be available by 2026–2027. |
| Q: Is hair loss normal on GLP-1 drugs? |
A: Hair shedding (telogen effluvium) is reported by some patients on GLP-1 drugs, particularly during periods of rapid weight loss. This is typically a physiological response to caloric restriction and rapid body weight changes — not a direct drug side effect. It is generally temporary and resolves as weight loss stabilizes. Adequate protein intake and nutritional support help minimize this effect. |
| Q: Can GLP-1 drugs help with PCOS? |
A: Polycystic ovary syndrome (PCOS) is closely linked to insulin resistance and metabolic dysfunction. Several small studies and clinical reports suggest GLP-1 drugs can improve menstrual regularity, ovulatory function, insulin sensitivity, and androgen levels in women with PCOS — particularly when weight reduction is achieved. Larger randomized trials are needed, and this remains an off-label use requiring specialist guidance. |
26. FUTURE TRENDS IN METABOLIC MEDICINE
The GLP-1 story is far from complete. The next decade promises a wave of innovation that will expand, refine, and deepen our ability to treat metabolic disease.
Next-Generation Molecules in Development
Oral GLP-1/GIP dual agonists (Orforglipron, Danuglipron) — once-daily pills with injectable-level efficacy
Triple agonists (GLP-1 + GIP + Glucagon) — Retatrutide showed up to 24% weight loss in Phase 2 trials (2023)
Monthly or quarterly injectable formulations for improved adherence
GLP-1-based combination therapies with amylin (cagrilintide) — REDEFINE trials showing up to 25%+ weight loss
Topical GLP-1 patches and implantable delivery systems under early investigation
Expanding Clinical Applications
Formal approvals expected for sleep apnea (tirzepatide SURMOUNT-OSA — positive results 2024)
Heart failure with preserved ejection fraction (HFpEF) — STEP-HFpEF trial positive
Alcohol use disorder (AUD) — Phase 2 trials actively recruiting
Non-alcoholic steatohepatitis (NASH) approvals imminent
Alzheimer's prevention trials completing 2026–2028
The Democratization of Metabolic Medicine
As patents expire and biosimilar GLP-1 medications enter the market (expected 2026–2031), costs are anticipated to decline substantially — potentially bringing these transformative medications to millions of patients currently priced out. Coupled with expanding telehealth infrastructure and improving insurance coverage advocacy, the future of metabolic medicine is one of increasing access and equity.
| 💡 LOOKING AHEAD |
| GLP-1 drugs may be remembered not merely as the weight-loss medications of the 2020s, but as the moment metabolic medicine grew up — when medicine stopped treating obesity's consequences and started treating its causes. The science is young, the applications are expanding, and the human potential is extraordinary. |
FINAL MEDICAL RESPONSIBILITY STATEMENT
| ⚠ EVIDENCE-BASED MEDICAL RESPONSIBILITY STATEMENT |
| All information contained in this article is provided in good faith for educational purposes only. Every effort has been made to present accurate, current, and evidence-based information consistent with peer-reviewed medical literature and guidance from regulatory authorities including the FDA, EMA, and major medical organizations. This content does NOT replace individualized medical advice from a licensed healthcare provider. Medical science evolves rapidly — verify all treatment decisions with your physician. Last reviewed: May 2026. |
Citation Placeholders:
- [1] Lincoff AM et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. NEJM. 2023.
- [2] Jastreboff AM et al. Tirzepatide Once Weekly for the Treatment of Obesity. NEJM. 2022.
- [3] Perkovic V et al. Semaglutide in Patients with Chronic Kidney Disease. NEJM. 2024.
- [4] Drucker DJ. The Biology of Incretin Hormones. Cell Metabolism. 2022.
- [5] Klausen MK et al. Exenatide reduces alcohol consumption in alcohol-dependent animals. JCI Insight. 2022.
- [6] ELAD Trial Investigators. Liraglutide in Alzheimer's Disease. Lancet. 2024.
- [7] FDA Drug Safety Communication. GLP-1 Receptor Agonist Review. FDA.gov. 2023.
- [8] WHO Medical Alert: Substandard/Falsified semaglutide. WHO.int. 2024.
